Aims and objectives
Tuberculosis (TB) remains the leading cause of death due to a single pathogen, Mycobacterium tuberculosis (Mtb; except in 2020-2022, when it was surpassed by the COVID-19 pandemic), with 1.5 million deaths in 2020. The lengthy TB treatment and the numerous adverse events contribute to poor medical adherence and development of antibiotic resistant strains. Thus, novel therapeutic modalities are urgently needed to shorten treatment duration, improve outcomes and control the emergence of drug resistant TB.
The ITHEMYC project convenes a multidisciplinary consortium of 11 partners, including two Product Development Partnerships (TBVI, TB-Alliance) and an industrial partner (GSK) involved in vaccine, drug and biomarker R&D for TB. The partners will work jointly to develop innovative adjunctive TB immunotherapies by capitalizing on a promising pipeline and recent developments in the field. The project will combine current and new antibiotic regimens with novel immunotherapies, such as small molecules targeting host pathogen-interactions, including host-directed therapies and virulence inhibitors, immunomodulatory compounds, monoclonal antibodies and therapeutic vaccines. The project will generate robust preclinical safety and efficacy information on compounds and combinations through a set of relevant in vitro, in vivo and in silico models, and progress two of them up to preclinical proof-of-concept in non-human primates within the project duration. The partners are proposing a critical path for characterization and progression of immunotherapies, that will be refined based on knowledge generated in ITHEMYC aiming to increase the understanding and interest for this emerging concept of adjunctive TB immunotherapy. We expect the new combined interventions will improve TB cure rates, reduce the duration and toxicity of current regimens and reduce relapse rates.
Work packages
ITHEMYC is divided into five work packages.
- WP1: Characterization of immunotherapies involved in innate response
- WP2: In vivo studies
- WP3: In silico modelling
- WP4: Management, stakeholder engagement and dissemination & exploitation
- WP5: Ethics requirements
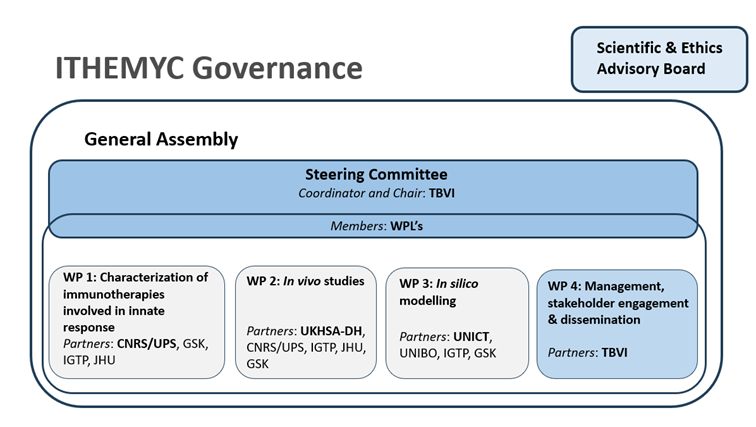
Governance
The ITHEMYC management structure was designed to optimally support this project and consists of a General Assembly (GA), WP Leaders (WPL), a Steering Committee (SC) and a Management Team (MT). The consortium will be advised by a Scientific and Ethics Advisory Board (SEAB).